M. Hähnel-Taguchi

Group leader, Margarete von Wrangell Fellow
University of Freiburg
Biology I, Developmental Biology
Hauptstraße 1, Room 2046
D-79104 Freiburg
Phone. ++49 / 761/203-2586
E-Mail: melanie.haehnel@biologie.uni-freiburg.de
Research Interests
How does the nervous system encode and process sensory information? What role does the modulation of sensory circuits play in adapting to different situations? How does sensory information transform into behavior? We are studying these questions in the zebrafish model system.
A five-day-old zebrafish larva already has a rich repertoire of behaviors that enable this small organism to avoid predators and survive. It uses visual, auditory and somatosensory cues, as well as information from the water motion, which it receives through the lateral line system, a unique sensory organ of fishes and aquatic amphibians. The receptors of the lateral line are mechanosensory hair cells, similar to those found in the auditory and vestibular system. Therefore, the study of the lateral line can provide insight into the general function of hair cell systems. The sensory circuitry of the lateral line receives neuromodulatory input at multiple levels. Neuromodulation can modify sensitivity and gain of a sensory circuit in the context of input from other sensory modalities, motor activity, or behavior.
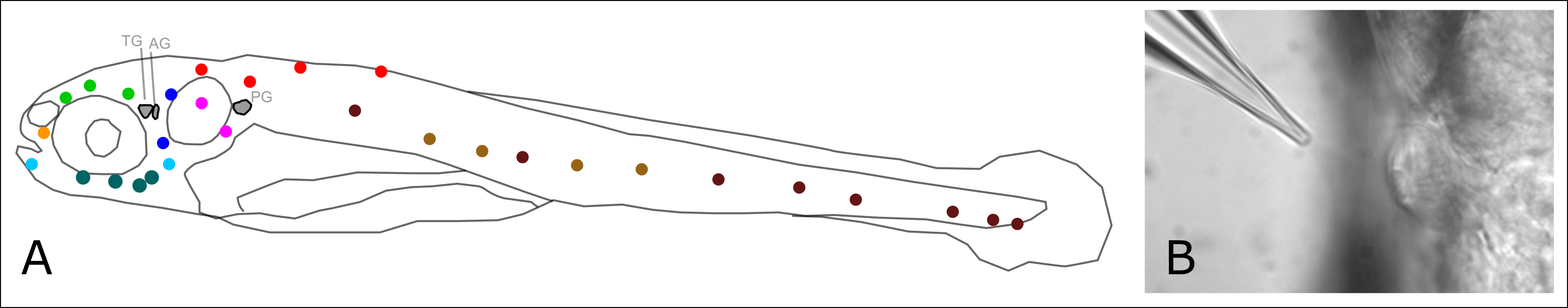
Figure1: The lateral line sensory Organ.
(A) Schematic of a 5-day-old zebrafish larva with neuromasts of the different branches of the lateral line depicted in different colors. TG: trigeminal ganglion, AG: anterior lateral line ganglion, PG: posterior lateral line ganglion.
(B) A neuromast can be stimulated with a water jet ejected from a micropipette, driven by a computer-controlled pump.
An important neuromodulator in the central nervous system is Dopamine. In zebrafish, a group of dopaminergic neurons in the diencephalon that corresponds to the A11 cluster in mammals, projects to multiple sensory systems at various levels and into the spinal cord. Also in the mammalian system, the A11 dopaminergic neurons are the only group that send descending projections into the spinal cord. They assume an important role in sensory modulation and abnormal dopamine signaling in these neurons plays a role in chronic pain and stands in connection to a common neurological disorder, the Restless Legs Syndrome (RLS), in which patients experience unpleasant sensation, mainly in the legs during rest.
Work from the Driever lab has shown that the far projecting diencephalic dopaminergic groups in the zebrafish depend on the transcription factor orthopeadia, and therefore correspond to the far projecting A11 neurons in mammals. It appears, due to their prominent innervation of the lateral line circuitry, and considering a recent study from the Driever lab (Reinig et al., 2017) which showed that activity in zebrafish A11-type neurons correlates with stimulation of the lateral line and the trigeminal system, this dopaminergic group is involved in the modulation of sensory processing in the zebrafish larva.
We are studying the zebrafish larval sensory circuits and their modulation using a combination of electrophysiology, imaging and behavioral experiments. Because of its small size and transparency, this model is ideally suited to conduct neurophysiological studies, and offers the opportunity to express fluorescent and calcium sensitive dyes in neuronal populations. The availability of many individuals of free-swimming, readily behaving larva also allows us to compare neurophysiological data to behavioral responses in free-swimming larvae with intact and perturbed dopaminergic signaling.
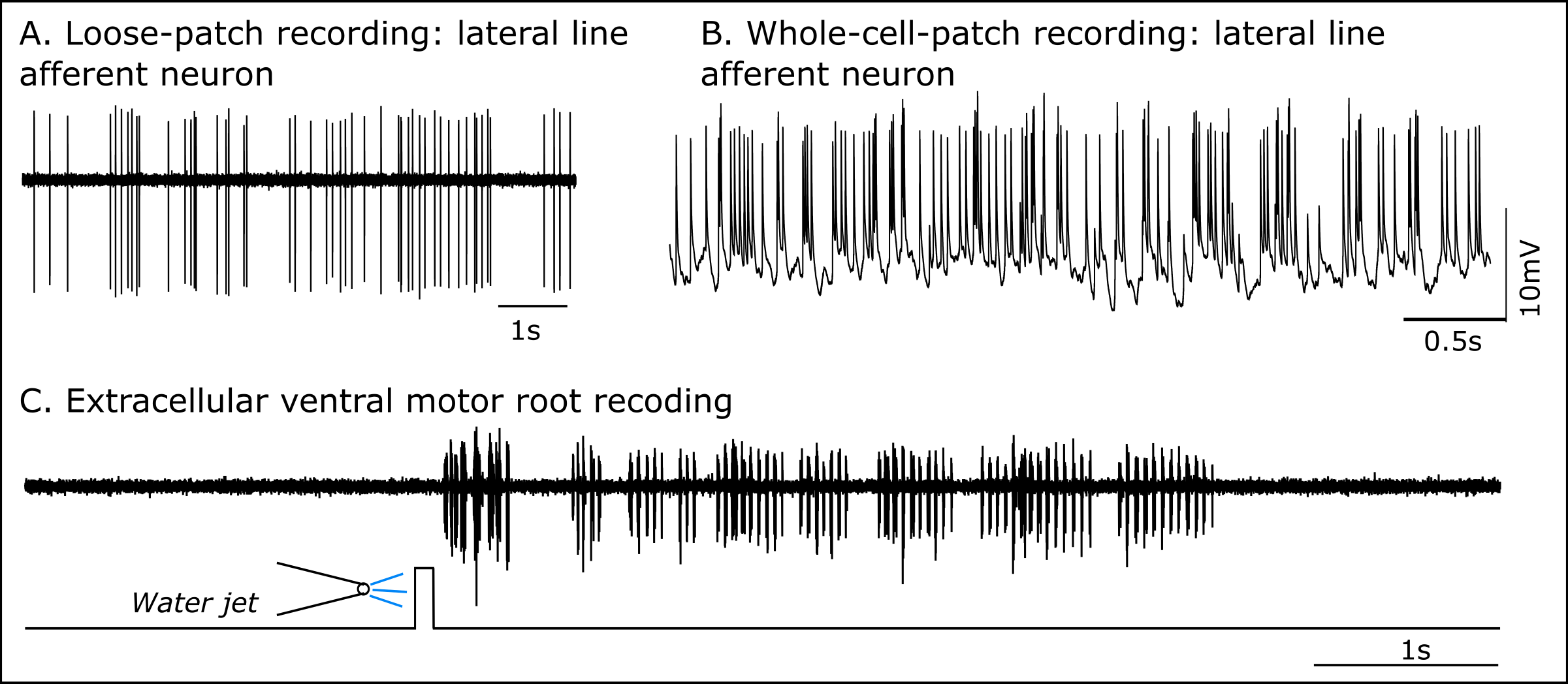
Figure 2: Electrophysiological recordings of larval zebrafish neurons
(A) Spontaneous activity of lateral line afferent neuron recorded in loose-patch configuration in the posterior lateral line ganglion.
(B) Spontaneous activity of lateral line afferent neuron recorded in whole cell-path configuration in the posterior lateral line ganglion.
(C) “Fictive swimming” activity of motor neurons in response to a computer controlled water jet directed towards the lateral line neuromasts, recorded extracellularly from the ventral motor root.
Students interested in a bachelor or master thesis regarding the topics described above, please contact me.
Short CV
- 01/2016 – present
Group leader at Developmental Biology Unit, University of Freiburg - 01/2015 – 08/2015
Adjunct teaching faculty, St. Johns River State College, Jacksonville, Florida - 05/2010 – 11/2015
Postdoctoral researcher in the Liao lab at Whitney Laboratory for Marine Bioscience, University of Florida - 03/2009 – 4/2010
Postdoctoral researcher in the Grünewald lab, Goethe University Frankfurt. - 2005 – 2009
Doctoral studies in the Menzel lab at Neurobiology Unit, Free University of Berlin. Dissertation: Characterization of mushroom body extrinsic neurons in the honeybee (Apis mellifera) and their role in learning and memory formation: a calcium imaging study. - 08/2005
Diploma Thesis in the DeBruyne lab at Neurobiology Unit, Free University of Berlin. Thesis title: Contribution from odor responses in individual olfactory sensilla to the electroantennogram in wildtype and mutant Drosophila melanogaster. - 1999 – 2005
Study of Biology, Free University of Berlin
Awards and Funding
- Since 03/2016
Margarete-von-Wrangell Fellowship (Habilitationsstipendium für Frauen des Landes Baden-Württemberg) - 2016
Sachmittelförderung der Wissenschaftlichen Gesellschaft Freiburg - 01/2012 – 12/2014
DFG Postdoc Stipendium
Publications
Peer-Reviewed Journal Articles:
- Haehnel-Taguchi M, Akanyeti O, Liao J (2014): Afferent and motor neuron activity in response to single neuromast stimulation in the posterior lateral line of larval zebrafish. Journal of Neurophysiology 112(6), 1329-1339.
- Olszewski J*, Haehnel M*, Taguchi M, Liao J (2012): Zebrafish larvae exhibit rheotaxis and detect and escape a continuous suction source using their lateral line. PLoS ONE 7(5), e36661 (*joint first authorship).
- Haehnel M, Menzel R (2012): Long-term memory and response generalization in mushroom body extrinsic neurons in the honeybee Apis mellifera. Journal of Experimental Biology 215(3), 559-565.
- Liao JC, Haehnel M (2012): Physiology of afferent neurons in larval zebrafish provides a functional framework for lateral line somatotopy. Journal of Neurophysiology 107(10), 2615-2623.
- Haehnel M, Taguchi M, Liao JC (2012): Heterogeneity and dynamics of lateral line afferent innervation during development in zebrafish (Danio rerio). The Journal of Comparative Neurology 520 (7), 1376-1386.
- Tom W, de Bruyne M, Haehnel M, Carlson JR, Ray A (2010): Disruption of olfactory receptor neuron patterning in Scutoid mutant Drosophila. Molecular and Cellular Neurosciences 46(1), 252-261.
- Haehnel M, Menzel R (2010) Sensory representation and learning-related plasticity in mushroom body extrinsic feedback neurons of the protocerebral tract. Frontiers in Systems Neuroscience 4.
- Haehnel M, Froese A, Menzel R (2009) In vivo Ca2+ imaging of mushroom body neurons during olfactory learning in the honey bee. Journal of Visualized Experiments: JOVE, doi: 10.3791/1353.
- Schmuker M, de Bruyne M, Hähnel M, Schneider G (2007) Predicting olfactory receptor neuron responses from odorant structure. Chemistry Central Journal 1(11).
Reports and Conference Proceedings:
- Smith CA, Haehnel-Taguchi M, Liao JC (2014) Regional Specialization of Posterior Lateral Line Efferent Neurons in the Hindbrain of Larval Zebrafish. Integrative and Comparative Biology 54, E351-E351.
- Liao JC, Akanyeti O, Ballo A, Haehnel M, Levi R (2014) Sensory and Motor Responses to Deflection of Single Neuromasts in the Lateral Line System in Larval Zebrafish. Integrative and Comparative Biology 54, E123-123.
- Kustermann T et al. (2010): Association of Institutes for Bee Research Report of the 57th Seminar in Bochum 23-25 March 2010. Apidologie 41 (6), 676-694.