PD Jörn Schweitzer

PD Privatdozent
University of Freiburg
Biology I, Developmental Biology
Hauptstraße 1
D-79104 Freiburg
Email: joern.schweitzer@biologie.uni-freiburg.de
Research interests
The establishment of functional neural connections requires the precise growth of axons to specific target areas. During development, axons are guided along specific pathways by attractive and repulsive guidance cues in the environment. Among the first axon pathways to be established in the embryonic vertebrate nervous system are longitudinal tracts. These tracts are of great importance, since they transmit all information between the brain and spinal cord. Abnormal formation of longitudinal axon tracts leads to severe behavioral defects in humans, such as congenital mirror movements, further underscoring their relevance.
However, the mechanisms regulating formation and guidance of longitudinal axons remain largely undefined. We are utilizing the zebrafish model system to decipher molecular mechanisms orchestrating formation of far-reaching longitudinal hypothalamo-spinal (HTS) axons. Transgenic lines to visualize HTS axons have been generated and are currently used to study HTS axon guidance (Movie and Fig. 1A).
Movie 1: Formation of longitudinal HTS projections between 22 and 26 hours post fertilization is shown. Longitudinal HTS growth cones migrate from their origin in the hypothalamus towards the spinal cord. The neurons express a membrane gfp under the control of the otpb promoter (otpb:gfp; Fujimoto et al., 2011; Schweitzer et al., 2013)
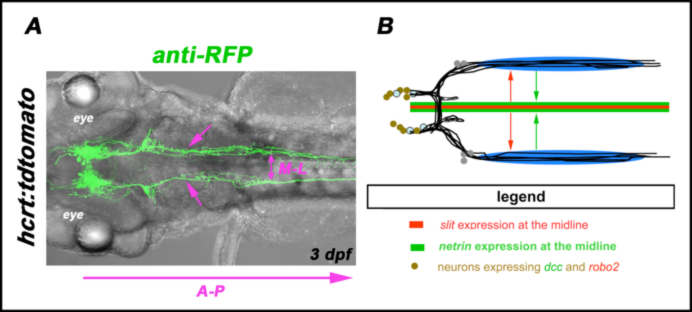
Work from our group demonstrated that simultaneous integration of repulsive Slit and attractive Netrin guidance signals from the ventral midline act in a concerted manner to define medio-lateral (M-L) positioning of longitudinal HTS axons (Fig. 1B; Kastenhuber et al., 2009, Kastenhuber et al., 2010 Schweitzer et al., 2011). Analyzing transcriptional control mechanisms of longitudinal HTS axon guidance we could show that longitudinal HTS growth cones ensure proper M-L positioning by regulating their responsiveness towards changing signaling intensities of guidance factors (Schweitzer et al., 2013). In addition to our work on M-L positioning we have now started to investigate molecular mechanisms underpinning longitudinal HTS axon guidance along the anterior-posterior (A-P) axis (Fig. 1A).
Future work in our group is directed to understand how far reaching longitudinal HTS growth cones modulate and integrate their responsiveness towards different signaling environments of guidance cues present at distinct steps of their journey. From a biomedical point of view, a better understanding of the development of long-distance connections between brain and spinal cord, such as longitudinal HTS axons may also provide insights for developing useful therapies to repair of these axons after injury.
Fig.2 Molecular mechanisms of longitudinal HTS axon guidance
(A) Dorsal view of a confocal z-projection of a hcrt:tdtomato transgenic embryo is shown. Anterior is left. Anti-RFP labeling revealed hypocretinergic neurons and their longitudinal HTS axons (arrows).
(B) Coordinated action of repulsion by Robo/Slit (red arrows) and attraction by DCC/Netrin (green arrows) orchestrate medio-lateral positionsing of longitudinal HTS axons (after Schweitzer et al., 2011). A-P: anterior-posterior; M-L medio-lateral; dpf: days post fertilization