Driever Lab
Prof. Dr. Wolfgang Driever
Hauptstrasse 1
D-79104 Freiburg
Phone: ++49 / 761 / 203 - 2587
E-Mail: driever@biologie.uni-freiburg.de
Research interests
Research in the laboratory focuses on the analysis of developmental mechanisms at the molecular level. We use the zebrafish as model system to combine genetics, genomics, signaling research, experimental embryology and optogenetics to achieve a quantitative understanding of complex signaling and regulatory networks in development. One focus is on nervous system development, aiming to understand neuronal differentiation as well as specific aspects of circuit formation and function.
Signaling and transcriptional networks controlling stem cells in the embryo
In the early embryo, stem cells are pluripotent and can form all tissues. However, later in development during organogenesis and nervous system development, tissue specific stem and progenitor cells give rise to defined cell types. We are interested in the signaling and transcriptional networks that control stem cell behavior. In the early embryo, the transcription factor Oct4 (Pou5f3 in zebrafish) is a major regulator of pluripotent cells. We investigated its functions during regulation of pluripotency network components and developmental control genes in the zebrafish blastula and gastrula. Our data have shown a fundamental role of Pou5f3 in temporal control of the early embryonic transcriptional networks (Onichtchouck et al. Mol. Sys. Biol. 2010). The large number of genes controlled by Pou5f3 can be explained by its evolutionarily conserved role in zygotic genome activation (Leichsenring et al., Science 2013). Pou5f3 does not only control transcriptional networks, but also cell behavior, adhesion and migration during gastrulation (Song et al., Developmental Cell 2013). Thus, the analysis of the embryo revealed potential mechanistic links between stem cell behavior in the embryo and cancer stem cell invasiveness.
In contrast to the broad potential of early embryonic stem cells, tissue-specific stem cells are located in defined signaling environments (called "stem cell niches") in the organs. We currently aim at understanding the control of a specific neural stem cell niche, and the transitions to progenitor cells and differentiating neurons in the zebrafish larval brain.
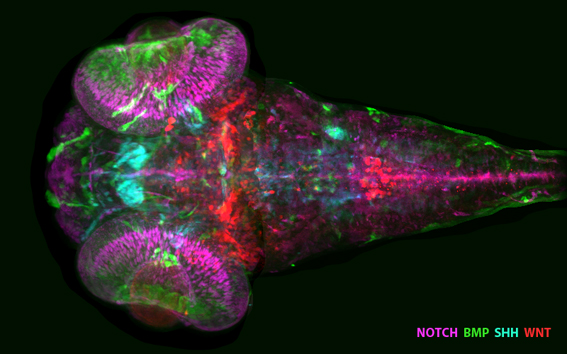
| A map of signaling activities in the developing zebrafish brain. The activities of the developmental signals BMP, SHH and WNT together with the neurogenesis signal of activated Notch are visualized using transgenic reporter lines expressing fluorescent proteins in response to specific signals. Confocal microscopy of three-day old larval zebrafish brains and image registration using our Vibe-Z system (Ronneberger et al., Nat. Meth. 2012) was used to generate this signaling map. Shown is a z-projection of a confocal stack, anterior is at left. |
Formation and function of dopaminergic systems
A biomedically relevant cell differentiation paradigm is the differentiation of dopaminergic neurons, which are affected in Parkinson's and other neurological diseases. These neurons use dopamine as transmitter to modulate the activities of other neurons involved in a broad range of behaviors. We investigate the signaling and transcriptional networks that control specification and differentiation of different dopaminergic neuron groups in the zebrafish embryo and larva (Filippi et al., Developmental Biology 2012). Using systems biology and virtualization tools, we aim to understand the complex networks of dopaminergic neuron differentiation (Ronneberger et al. Nature Methods 2012). Our interest extends into mechanisms to generate functional axonal connections of dopaminergic neurons to their targets (Tay et al. Nature Communications. 2011; Schweitzer et al., Development 2013). Zebrafish are an excellent model to link development and function of dopaminergic neuromodulation to control of behavior (Arrenberg and Driever, Frontiers Neurosciences 2013). For the so-called diencephalospinal dopaminergic system (homologous to A11 in mammals), we were able to demonstrate that its activity is tuned to sensory stimuli, and that distinct subgroups of neurons react to tactile or visual stimuli (Reinig et al., Current Biology 2017). We aim at an integrated understanding of differentiation of these neurons from stem cells, and their integration into functional circuits, to identify the genetic and molecular codes that specify nervous system function.
(PubMed links for references listed in text see Publications)
Membership in Research Networks